Hire Verified & Experienced
Pharmacovigilance Tutors
4.8/5 40K+ session ratings collected on the MEB platform
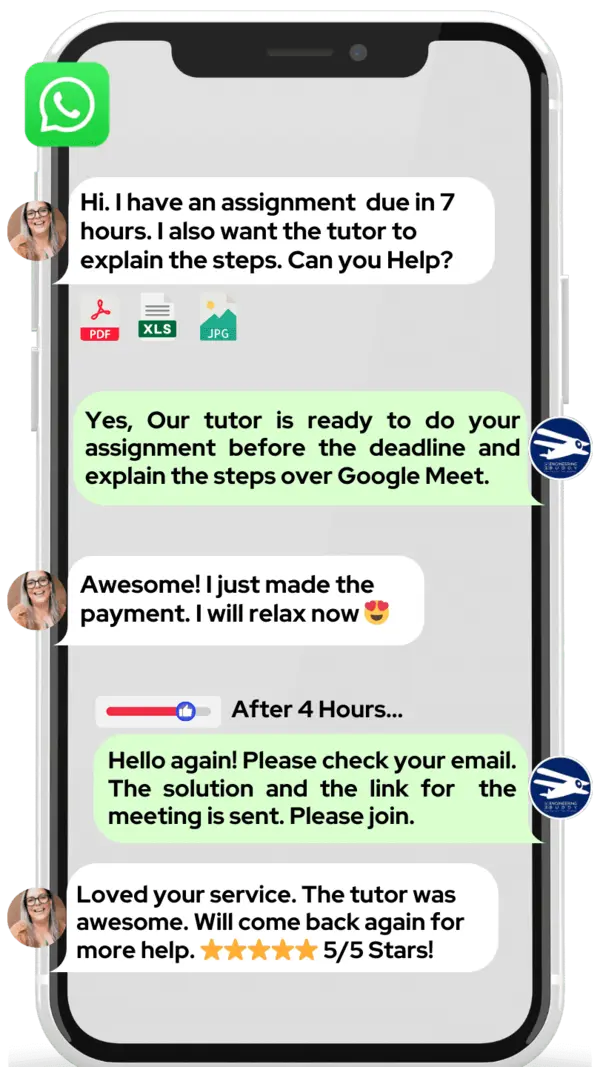
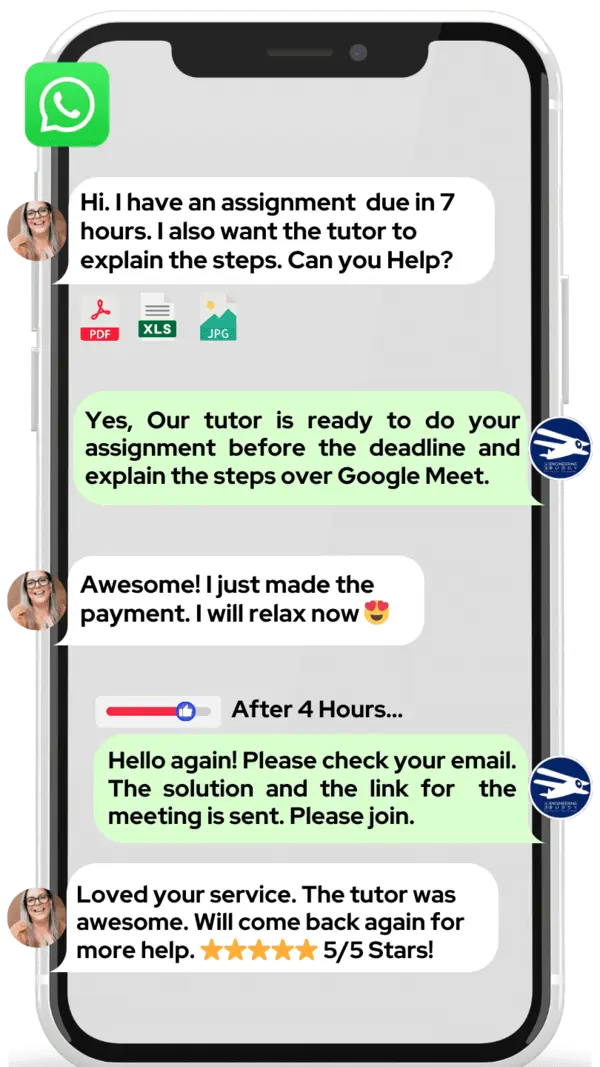
Hire The Best Pharmacovigilance Tutor
Top Tutors, Top Grades. Without The Stress!
52,000+ Happy Students From Various Universities
How Much For Private 1:1 Tutoring & Hw Help?
Private 1:1 Tutoring and HW help Cost $20 – 35 per hour* on average.
Most students who struggle with Pharmacovigilance aren’t short on effort — they’re short on someone who can explain adverse event causality assessment at 11 pm before a submission deadline.
Pharmacovigilance Tutor Online
Pharmacovigilance is the science of detecting, assessing, and preventing adverse drug reactions. Governed by ICH E2 guidelines and regulated by agencies such as the EMA and FDA, it equips practitioners to ensure medicine safety post-approval.
MEB connects you with a qualified Pharmacovigilance tutor online who knows ICH E2A–E2F, signal detection, ICSR reporting, and risk management planning — not just the textbook definitions, but the assessment logic behind them. If you’ve searched for a Pharmacovigilance tutor near me, working live online with a specialist is faster and more flexible. Our Pharmacology tutoring platform has served students across the US, UK, Canada, Australia, and the Gulf since 2008. One clear outcome: students leave sessions able to apply regulatory logic to real case scenarios — not just recall definitions.
- 1:1 online sessions tailored to your course syllabus and regulatory framework
- Expert-verified tutors with backgrounds in drug safety, clinical pharmacology, or regulatory affairs
- Flexible time zones — US, UK, Canada, Australia, Gulf covered
- Structured learning plan built after a diagnostic session
- Ethical homework and assignment guidance — you understand the work before you submit
52,000+ students across the US, UK, Canada, Australia, and the Gulf have used MEB since 2008 — including students in Pharmacology subjects like Pharmacovigilance, Pharmacokinetics, and Pharmacogenomics.
Source: My Engineering Buddy, 2008–2025.
How Much Does a Pharmacovigilance Tutor Cost?
Most Pharmacovigilance sessions run $20–$40/hr depending on level and topic depth — ICSR narrative writing and signal detection modules tend to sit at the higher end. New students can start with the $1 trial before committing to anything.
| Level / Need | Typical Rate | What’s Included |
|---|---|---|
| Standard (most levels) | $20–$35/hr | 1:1 sessions, homework guidance |
| Advanced / Specialist | $35–$70/hr | Expert tutor, regulatory depth, niche topics |
| $1 Trial | $1 flat | 30 min live session or 1 homework question explained |
Tutor availability tightens significantly around university coursework deadlines and pharmaceutical regulatory exam cycles — book early if you have a fixed date.
WhatsApp MEB for a quick quote — average response time under 1 minute.
Who This Pharmacovigilance Tutoring Is For
This covers undergraduate pharmacy and life sciences students, postgraduate students on MSc Drug Safety or Regulatory Affairs programmes, and industry professionals upskilling for roles at pharmaceutical companies, CROs, or regulatory agencies. If you’re at universities such as King’s College London, Monash University, Utrecht University, University of Toronto, or Georgetown University, MEB tutors are familiar with programme structures at this level.
- Undergraduate pharmacy students working through adverse drug reaction modules for the first time
- MSc or graduate students whose signal detection or risk benefit assessment coursework is due within weeks
- Students retaking after a failed first attempt — particularly those who struggled with causality assessment frameworks or ICSR narrative structure
- Professionals transitioning into drug safety roles who need to close gaps in ICH E2E or E2R knowledge fast
- Students who need guided homework and assignment support — you do the work; the tutor makes sure you understand it first
The $1 trial is a low-risk way to check the tutor’s fit before booking any regular sessions.
1:1 Tutoring vs Self-Study vs AI vs YouTube vs Online Courses
Self-study works if you’re disciplined, but Pharmacovigilance case analysis — particularly causality assessment using WHO-UMC or Naranjo criteria — gives most students real trouble without feedback. AI tools explain terminology fast, but can’t assess whether your ICSR narrative logic is actually sound. YouTube covers concepts at a surface level and stops short when you need to work through a specific adverse event scenario. Online courses follow a fixed structure regardless of where your gaps are. 1:1 tutoring with MEB is live, responds to your exact coursework or exam question, and corrects regulatory reasoning errors in real time.
Outcomes: What You’ll Be Able To Do in Pharmacovigilance
After working with an MEB Pharmacovigilance tutor, students can apply causality assessment criteria — including Naranjo and WHO-UMC scales — to real adverse event reports with confidence. You’ll be able to write ICSR narratives that meet E2B(R3) formatting requirements, analyze signal detection data from sources like VigiBase or the FDA Adverse Event Reporting System, explain the components of a Risk Management Plan under EMA guidelines, and present benefit-risk evaluations using structured frameworks used in regulatory submissions.
Based on feedback from 40,000+ sessions collected by MEB from 2022 to 2025, 58% of students improved by one full grade after approximately 20 hours of 1:1 tutoring in subjects like Pharmacovigilance. A further 23% achieved at least a half-grade improvement.
Source: MEB session feedback data, 2022–2025.
What We Cover in Pharmacovigilance (Syllabus / Topics)
Regulatory Frameworks and ICSR Reporting
- ICH E2A–E2F guidelines: scope, definitions, and reporting obligations
- Individual Case Safety Report (ICSR) structure under E2B(R3)
- MedDRA coding: SOC, HLGT, HLT, PT, LLT hierarchy and selection logic
- EMA, FDA, MHRA, and Health Canada reporting timelines and requirements
- Expedited vs periodic reporting: 15-day rules and PSUR/PBRER structure
- Data privacy considerations in ICSR submissions (GDPR and equivalents)
Recommended texts: Stephens’ Detection of New Adverse Drug Reactions (Talbot & Aronson); A Practical Guide to Pharmacovigilance (Dave & Bhatt). Core ICH guideline documents from the official ICH website are essential alongside any textbook.
Causality Assessment and Signal Detection
- Causality assessment methods: Naranjo algorithm, WHO-UMC criteria, Bradford Hill
- Imputability: certain, probable, possible, unlikely — applying criteria to case narratives
- Signal detection methods: disproportionality analysis, PRR, ROR, BCPNN
- Data mining in pharmacovigilance databases: VigiBase, FAERS, EudraVigilance
- Signal evaluation, prioritisation, and escalation workflows
- Case study application: working through real-world signal detection scenarios
Recommended texts: Signal Detection Methods in Pharmacovigilance (Hauben & Bate); Pharmacovigilance: An International Textbook (Mann & Andrews).
Risk Management and Benefit-Risk Evaluation
- Risk Management Plans (RMP): EMA template structure and content requirements
- Risk Minimisation Measures (RMM): routine and additional measures
- Benefit-risk assessment frameworks: BRAT methodology and structured templates
- Periodic Safety Update Reports (PSUR) and PBRER format and content
- Post-marketing surveillance study design and outcomes reporting
- Communication of drug safety information: DHPC, label updates, and Dear Healthcare Professional letters
Recommended texts: Risk Management in Pharmacovigilance (Suvarna); Benefit-Risk Appraisal of Medicines (Hauben & Reich). The NIH MedlinePlus database is a useful adjunct for verifying drug-specific safety profiles during case analysis.
What a Typical Pharmacovigilance Session Looks Like
The tutor starts by checking the previous topic — often causality assessment criteria or MedDRA coding logic — asking the student to walk through a specific case scenario they attempted between sessions. From there, student and tutor work through a live ICSR narrative together on screen: the tutor uses a digital pen-pad to annotate the E2B(R3) fields while the student explains their coding choices. If signal detection is on the agenda, they’ll run through a PRR or ROR calculation step by step before the student replicates the method independently. The session closes with a specific task — drafting a benefit-risk summary section or working through three causality assessment cases — and the next topic logged for follow-up.
How MEB Tutors Help You with Pharmacovigilance (The Learning Loop)
Diagnose: In the first session, the tutor identifies exactly where the breakdown happens — is it the regulatory hierarchy, the narrative structure of an ICSR, the statistical logic behind signal detection, or the policy framework of a Risk Management Plan? Most students have one or two specific gaps, not a wholesale knowledge problem.
Explain: The tutor works through live examples on screen — building an ICSR narrative field by field, or walking through a Naranjo algorithm assessment on a real case. The digital pen-pad makes every step visible and traceable.
Practice: The student attempts the next case or question with the tutor present. No moving on until the reasoning is sound, not just the answer.
At MEB, we’ve found that Pharmacovigilance students who struggle with causality assessment are rarely confused about the criteria themselves — they’re applying them mechanically without reading the clinical narrative first. One session focused on that sequence changes everything.
Feedback: The tutor works through errors step by step — not just marking something wrong, but showing exactly where the regulatory logic broke down and why that would cost marks or fail an ICSR quality check.
Plan: After each session, the tutor logs the next topic in sequence and sets a specific task. If an exam or submission deadline is approaching, the session plan is compressed accordingly.
Sessions run on Google Meet. The tutor uses a digital pen-pad or iPad with Apple Pencil. Before your first session, share your course syllabus or exam board, a recent assignment or past question you struggled with, and your deadline or exam date. The first session covers diagnosis and the first substantive topic — you won’t spend the hour on admin. Whether you need a quick catch-up before a coursework deadline, structured revision over four to eight weeks, or ongoing weekly support through a semester-long Drug Safety module, the tutor maps the session plan after that first diagnostic.
Start with the $1 trial — 30 minutes of live tutoring that also serves as your first diagnostic.
Students consistently tell us that the hardest part of Pharmacovigilance isn’t the volume of content — it’s knowing which regulatory framework applies to which scenario. That’s exactly what a 1:1 session can resolve in one sitting.
Tutor Match Criteria (How We Pick Your Tutor)
MEB matches tutors based on four factors, not just subject name.
Subject depth: The tutor must have worked at the level of your course — undergraduate pharmacy safety modules, MSc Drug Regulatory Affairs, or professional upskilling for CRO or pharmaceutical company roles — not just general pharmacology knowledge.
Tools: Every session uses Google Meet with a digital pen-pad or iPad and Apple Pencil — essential for annotating ICSR forms, signal detection tables, and regulatory templates live on screen.
Time zone: Matched to your region — US, UK, Gulf, Canada, or Australia — so sessions run at hours that don’t require you to be awake at 3 am.
Goals: Whether you need exam score improvement, conceptual clarity on benefit-risk frameworks, or pharmaceutical homework help, the tutor is selected against that specific goal.
Unlike platforms where you fill out a form and wait, MEB responds in under a minute, 24/7. Tutor match takes under an hour. The $1 trial means you test before you commit. Everything runs over WhatsApp — no logins, no intake forms.
Pricing Guide
Rates run $20–$40/hr for most Pharmacovigilance coursework — undergraduate adverse drug reaction modules, PSUR structure, and ICSR reporting typically fall here. Signal detection methodology and RMP writing at postgraduate or professional level can reach $70–$100/hr depending on tutor background.
Rate factors: your level, the complexity of the specific topic, your timeline, and tutor availability at your preferred hours.
Demand is high around university assessment periods in March–April and October–November. If your deadline is fixed, book sooner rather than later.
For students targeting roles at major pharmaceutical companies, EMA, MHRA, or preparing for the Drug Safety qualification from DIA or TOPRA, tutors with direct regulatory affairs or pharmacovigilance industry experience are available at higher rates — share your specific goal and MEB will match the tier.
Start with the $1 trial — 30 minutes, no registration, no commitment. WhatsApp MEB for a quick quote.
MEB has matched tutors across 2,800+ subjects since 2008 — from undergraduate Pharmacovigilance modules to postgraduate Drug Safety programmes — with a response time averaging under one minute via WhatsApp.
Source: My Engineering Buddy, 2008–2025.
FAQ
Is Pharmacovigilance hard?
Yes — the regulatory detail is dense and the application logic is unforgiving. Students who struggle most are typically strong on memorised definitions but weak on applying causality criteria or MedDRA coding logic to actual case narratives. That gap closes faster with a tutor than alone.
How many sessions are needed?
Two to three sessions resolve most single-topic gaps — ICSR narrative structure or a specific causality method. Broader support across a full Drug Safety module typically runs eight to twelve sessions over a semester, depending on starting level and assessment schedule.
Can you help with homework and assignments?
MEB tutoring is guided learning — you understand the work, then submit it yourself. The tutor explains the regulatory logic behind your assignment question, walks through the method, and checks your reasoning — not your final submission. See our Academic Integrity policy and Why MEB page for full details on what we help with and what we don’t.
Will the tutor match my exact syllabus or exam board?
Yes. Before the first session, share your course outline, institution, and assessment format. MEB tutors are matched to your specific content — ICH guidelines, EMA or FDA focus, PSUR vs PBRER requirements — not to a generic Pharmacovigilance curriculum.
What happens in the first session?
The tutor runs a short diagnostic — usually a case-based question or a past assignment you struggled with — to identify exactly where the reasoning breaks down. The session then moves immediately into the first substantive topic. No time is spent on admin or generic introductions.
Is online tutoring as effective as in-person?
For Pharmacovigilance, yes — and often better. Regulatory documents, ICSR forms, signal detection tables, and MedDRA coding hierarchies are all screen-based. Working through them live on Google Meet with a digital pen-pad is closer to real regulatory work than any whiteboard session.
What is the difference between ICH E2A and E2B in Pharmacovigilance, and do I need to know both?
E2A defines clinical safety data definitions and reporting timelines for expedited reports. E2B(R3) specifies the electronic transmission format for ICSRs. Both appear in most postgraduate and professional assessments — your tutor covers whichever your course emphasises, or both if needed.
How do MEB tutors approach causality assessment — do they teach one method or all of them?
Tutors teach the method your course specifies first — Naranjo, WHO-UMC, or Bradford Hill — then build comparative understanding so you can explain why one framework applies in a given regulatory context. Examiners often test that distinction directly.
Can I get Pharmacovigilance help at midnight?
Yes. MEB operates 24/7 across time zones. Students in the Gulf, Australia, and the US West Coast regularly book late-night sessions. WhatsApp MEB at any hour and a tutor is typically matched within 60 minutes.
What if I don’t like my assigned tutor?
Request a switch via WhatsApp — no forms, no delay. MEB re-matches you at no additional charge. The $1 trial exists precisely so you can assess fit before committing to ongoing sessions.
How do I get started?
Three steps: WhatsApp MEB, get matched with a verified Pharmacovigilance tutor — usually within the hour — then start the $1 trial: 30 minutes of live 1:1 tutoring or one homework question explained in full. No registration required.
Trust & Quality at My Engineering Buddy
Every MEB tutor goes through subject-specific screening — not a generic interview. For Pharmacovigilance, that means verifying depth in ICH guidelines, signal detection methods, and ICSR regulatory logic, not just a pharmacology degree. Tutors complete a live demo evaluation and undergo ongoing session feedback review. Rated 4.8/5 across 40,000+ verified reviews on Google.
MEB tutoring is guided learning — you understand the work, then submit it yourself. For full details on what we help with and what we don’t, read our Academic Integrity policy and Why MEB.
MEB has served 52,000+ students across the US, UK, Canada, Australia, the Gulf, and Europe since 2008 — across 2,800+ subjects. In Pharmacology, that includes students working on Pharmacokinetics tutoring, Pharmacogenomics help, and Drug Discovery and Development — alongside Pharmacovigilance. See our tutoring methodology for how sessions are structured across all levels.
A common pattern our tutors observe is that students who arrive confident in regulatory theory become unsure the moment a case requires them to choose between two valid causality frameworks. That’s not a knowledge gap — it’s a decision-making gap. It’s fixable in one targeted session.
Explore Related Subjects
Students studying Pharmacovigilance often also need support in:
Next Steps
Ready to get started? Here’s what to do:
- Share your course outline or exam board, the specific topic where you’re stuck, and your deadline or exam date
- Share your availability and time zone — sessions are matched to your hours
- MEB matches you with a verified Pharmacovigilance tutor — usually within 24 hours, often within the hour
- The first session starts with a diagnostic so every minute is used well
Before your first session, have ready:
- Your course syllabus or module guide (ICH guidelines focus, EMA or FDA context)
- A recent assignment or past question you struggled with — ICSR narrative, causality assessment, or signal detection
- Your exam or submission deadline date
The tutor handles the rest. Visit www.myengineeringbuddy.com for more on how MEB works.
WhatsApp to get started or email meb@myengineeringbuddy.com.
Our experience across thousands of sessions shows that the students who improve fastest in Pharmacovigilance are those who arrive with a specific question — not a vague feeling of being lost. A concrete case, a past paper question, a single regulation that doesn’t make sense. Start there.
Reviewed by Subject Expert
This page has been carefully reviewed and validated by our subject expert to ensure accuracy and relevance.












