Mastering clinical research requires understanding complex science, ethics, and regulations. If you’re exploring programs such as the Radiologic Technology programs, specialized tutoring can bridge knowledge gaps, helping you navigate study design, data management and publication processes.

The Hidden World of Medical Breakthroughs: More Than Just a Lab Coat
Imagine this: a student named Sarah, bright and fascinated by medicine, staring at a diagram of a Phase III clinical trial. The chart is a labyrinth of acronyms, ethical checkpoints, and statistical jargon. She understands the biology, the chemistry, the “what” of the disease, but the “how” of developing a new treatment feels like an insurmountable wall. She’s not alone. Every year, thousands of students dive into health sciences, dreaming of making a difference, only to find themselves navigating the incredibly complex world of clinical research.
This isn’t just about memorizing facts from a textbook. It’s about understanding the very engine of medical progress. Clinical research is the bridge between a brilliant idea in a lab and a life-saving medicine on a pharmacy shelf. It’s a field that’s both intellectually thrilling and profoundly human. But let’s be honest, it can be tough. Really tough.
This article is for students like Sarah, and for the parents and teachers who support them. We’ll demystify this challenging subject, explore why it’s so demanding, and show how the right guidance can transform a struggling student into a confident future leader in healthcare innovation.
What Exactly Is Clinical Research?
At its heart, clinical research is the study of human health and illness. Scientists and doctors conduct studies with people to understand how to prevent, diagnose, and treat diseases. It’s the final, crucial step before a new drug, medical device, or procedure can be approved for public use.
Think of it as a meticulously planned journey with several distinct stages, known as phases:
- Phase I: The first time a new drug is tested in a small group of healthy volunteers to evaluate its safety, determine a safe dosage range, and identify side effects.
- Phase II: The drug is given to a larger group of people who have the disease to see if it is effective and to further evaluate its safety.
- Phase III: The treatment is given to large groups of people to confirm its effectiveness, monitor side effects, compare it to commonly used treatments, and collect information that will allow it to be used safely. The U.S. Food and Drug Administration (FDA) typically requires a successful Phase III trial before approving a new drug.
- Phase IV: These are post-marketing studies that gather additional information on the drug’s risks, benefits, and optimal use after it has been approved and is on the market.
It can be difficult to visualize how a drug moves from a lab concept to your local pharmacy shelf. This timeline breaks down the four critical phases of the clinical trial lifecycle.

Understanding the specific goal of each phase—from safety to efficacy—is the first step in mastering clinical research.
Notice how the focus shifts from safety in Phase I to long-term monitoring in Phase IV—this progression is the backbone of all clinical studies.
The scale of this endeavor is massive. As of 2023, there were over 437,000 registered clinical studies in 221 countries listed on ClinicalTrials.gov, a database managed by the U.S. National Library of Medicine. This highlights the global importance and the sheer volume of work involved in modern medicine.
Effective Clinical Research teaching focuses not just on these phases but on the intricate web of ethics, patient safety, and regulatory oversight that governs every single step. It’s a discipline where scientific rigor meets profound ethical responsibility.

Why Is Clinical Research So Hard to Grasp?
Many students struggle because they treat this field like general biology, but it is actually a complex intersection of four distinct professions. This diagram illustrates why a multidisciplinary approach is required.
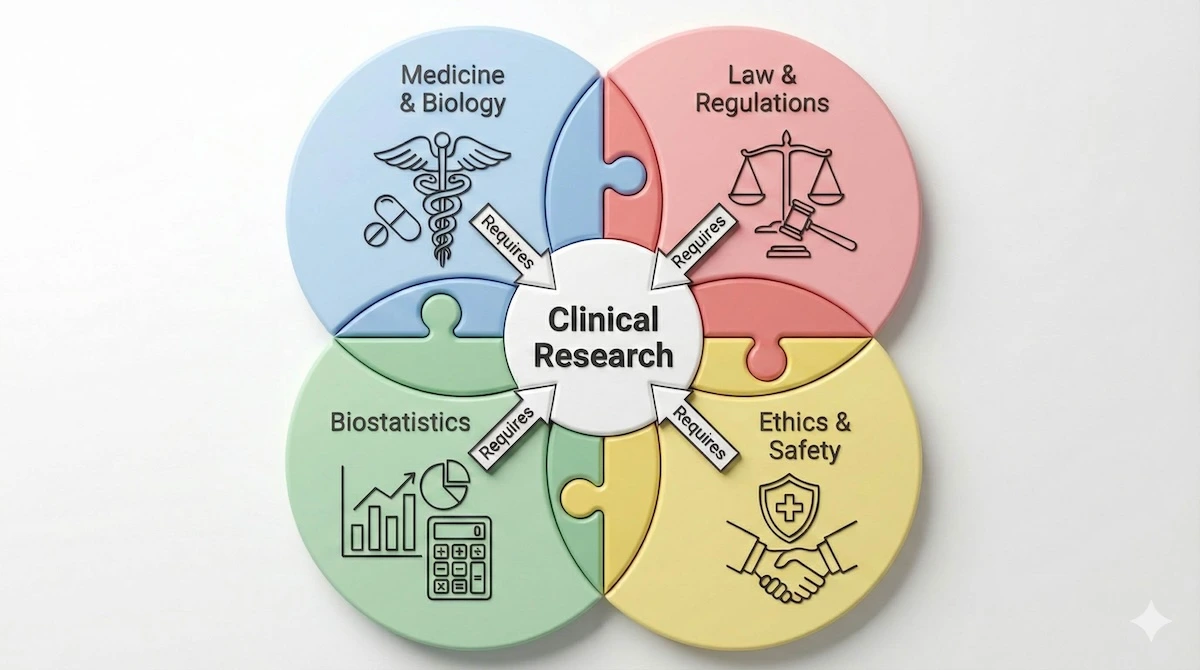
Clinical research isn’t just biology; it’s the complex intersection of four distinct professional fields.
To succeed, you cannot just master the science; you must also understand how the legal, ethical, and statistical components overlap.
Students often find this subject more challenging than standard biology or chemistry for several reasons. It’s not just one subject; it’s a multidisciplinary field that demands a unique combination of skills.
It’s a Mix of Many Subjects
Clinical research sits at the intersection of medicine, biostatistics, ethics, and law. One day you’re studying the pharmacology of a new compound, and the next you’re dissecting the legal implications of the Declaration of Helsinki. This requires mental agility and the ability to connect disparate fields of knowledge, a common reason students seek Clinical Research homework help.
The Language is Dense and Full of Acronyms
GCP (Good Clinical Practice), IRB (Institutional Review Board), FDA (Food and Drug Administration), CRO (Contract Research Organization)—the list of acronyms is endless. The terminology is precise and unforgiving. Misunderstanding a single term can change the meaning of an entire protocol. It’s like learning a new language, one where mistakes can have serious consequences.
Feeling lost in the alphabet soup? Use this quick reference guide to decode the most common acronyms you will encounter in your studies.

Save this “cheat sheet” to navigate the dense alphabet soup of industry terminology.
Mastering these core terms (like GCP and IRB) is the first step toward speaking the language of the industry fluently.
Ethical Considerations are Paramount
This is perhaps the most crucial and complex part of the field. The history of clinical research has dark chapters, leading to the development of strict ethical guidelines to protect human participants. Understanding the nuances of informed consent, patient confidentiality, and the ethical balance of risk versus benefit is non-negotiable. This abstract, high-stakes thinking can be a major hurdle for many learners.
Statistics are Everywhere
How do you know if a drug actually works? The answer lies in biostatistics. Students need to understand study design, sample size calculations, p-values, and data analysis to critically evaluate research findings. For many who excel in qualitative science, the heavy reliance on statistics can feel like a sudden detour into a math-heavy landscape, often leading them to seek Clinical Research assignment help.
The Digital Shift: Finding Your Mentor Online
Just as medicine has evolved, so has education. The image of a tutor is no longer limited to someone sitting across from you at a library table. The digital age has brought expertise to our fingertips. For a specialized field like this, online tutoring has been a game-changer.
Let’s go back to Sarah. Overwhelmed, she mentioned her struggles to a professor, who suggested she find a mentor who had worked in the field. Sarah’s search led her to the world of online tutoring, where she could connect with experts from around the globe.
This is where finding a dedicated Clinical Research tutor online can make all the difference. Instead of a general science tutor, you can learn from someone who has actually managed clinical trials, written study protocols, or worked for a pharmaceutical company. They’ve been in the trenches and can offer insights that go far beyond any textbook.
The beauty of Clinical Research tutoring online is its flexibility. Whether you’re a high school student in the UK exploring career options, a college student in the US struggling with a specific course, or a postgraduate in the Gulf countries working on a dissertation, you can access top-tier help that fits your schedule.
How a Great Tutor Can Change Everything
A good tutor doesn’t just give you answers. A great Clinical Research tutor builds a bridge between what you know and what you need to learn.
Consider the story of Mark, a university student who was on the verge of dropping his clinical sciences module. He just couldn’t connect the theory to practice. His grades were slipping, and his confidence was shattered. He decided to hire a Clinical Research tutor as a last resort. His tutor wasn’t just a teacher; he was a retired clinical trial manager. He didn’t just explain the phases of a trial; he shared stories of real-world challenges—of recruitment struggles, unexpected side effects, and the elation of a successful outcome.
Suddenly, the jargon clicked into place. The ethical dilemmas became real. The statistics told a story. The tutor provided personalized Clinical Research tutoring that was tailored to Mark’s learning style. He used real-world case studies to make complex topics relatable. Mark didn’t just pass the course; he aced it. More importantly, he regained his passion for the subject and is now pursuing a career in clinical data management.
You might wonder if a standard science tutor is enough. The table below compares general academic help with the specialized industry mentorship we provide.
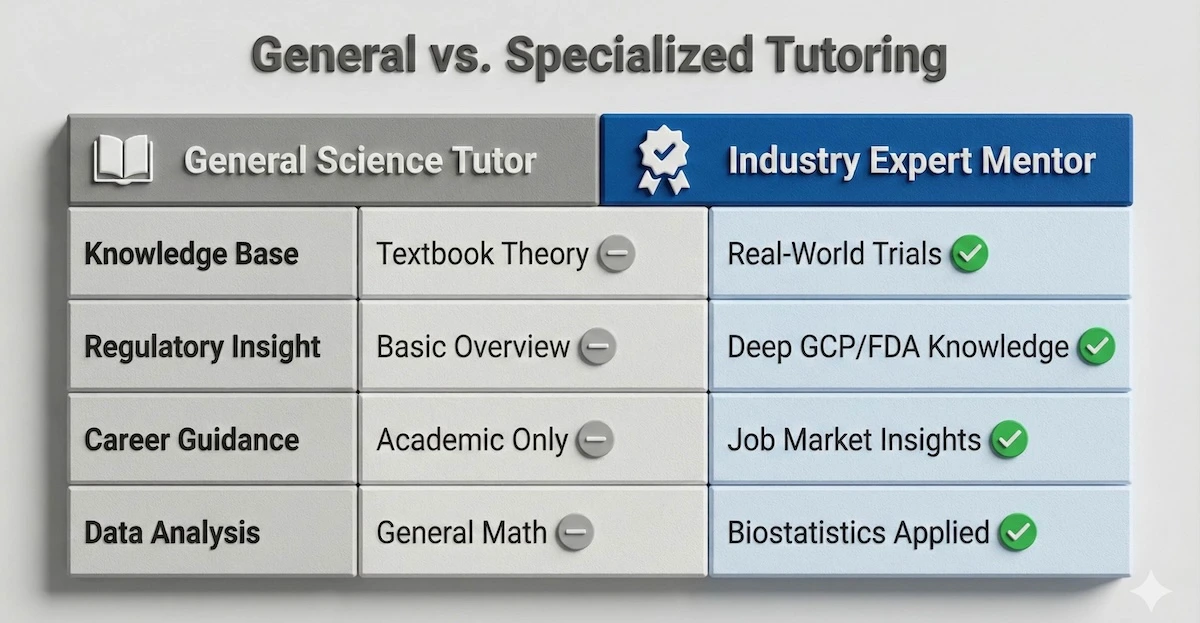
See the difference a specialized industry mentor makes compared to general academic help.
As you can see, real-world regulatory insight is the key difference that turns a student into a job-ready candidate.
This is the transformative power of one-on-one guidance. A skilled Clinical Research teacher can:
- Demystify Complex Topics: Break down intimidating subjects like regulatory affairs and biostatistics into manageable pieces.
- Provide Real-World Context: Share industry experience to make theoretical knowledge stick.
- Offer Personalized Support: Focus on your specific areas of weakness, whether it’s understanding protocols or analyzing data. This is especially helpful for targeted Clinical Research hw help.
- Boost Confidence: Create a safe space to ask questions you might be too embarrassed to ask in a large lecture hall.
Paving the Way for a Fulfilling Career
Mastering this subject is not just about getting a good grade; it’s about opening the door to a career that is both in-demand and incredibly rewarding. The field of clinical research is booming. The U.S. Bureau of Labor Statistics projects that employment for medical scientists, which includes many clinical research professionals, will grow 10 percent from 2022 to 2032, much faster than the average for all occupations.
Where can this knowledge take you? This career roadmap maps out your potential trajectory from student to leadership roles.
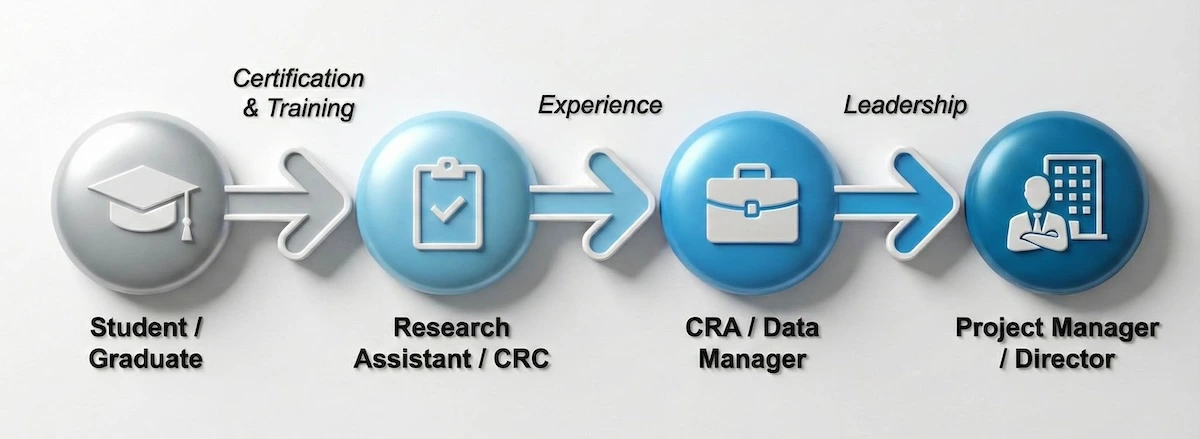
Your journey starts with education, but leads to diverse and high-growth leadership roles.
Whether you aim to be a Clinical Research Associate (CRA) or a Project Manager, the foundation you build today determines how fast you climb this ladder.
A strong academic foundation is the first step. Whether your goal is to become a Clinical Research Associate (CRA), a data manager, a regulatory affairs specialist, or a principal investigator, a deep understanding of the fundamentals is essential. This is where you need to start research into your own learning needs.
Don’t let complex terminology or daunting statistics stand in the way of your ambition. Getting help isn’t a sign of weakness; it’s a sign of commitment to your own success. The support of an expert can be the catalyst that transforms a challenging subject into a lifelong passion and a successful career.
Frequently Asked Questions (FAQs)
Q1. What is the main goal of clinical research?
The main goal is to determine the safety and effectiveness of new medical treatments and diagnostic methods in humans.
Q2. Why are ethics so important in this field?
Strict ethics are essential to protect the rights, safety, and well-being of the human volunteers who participate in studies.
Q3. Can a tutor help if I’m just bad at statistics?
Yes, a good tutor can focus on biostatistics, using real medical examples to make the concepts easier to understand and apply.
Q4. Is online tutoring as effective as in-person?
For specialized subjects like this, online tutoring is often more effective as it gives you access to a wider pool of true industry experts.
Q5. What kind of jobs can I get with a background in clinical research?
Careers include roles like Clinical Research Coordinator, Associate, data manager, and regulatory affairs specialist in hospitals, pharma companies, and government agencies.
Q6. When is the best time to get a tutor for this subject?
The best time is as soon as you start feeling overwhelmed, as the concepts build upon each other and early intervention is key.
Related Subjects
Internal Medicine
Medical Coding
Physical Therapy (Physiotherapy)
Speech-Language Pathology and Audiology
******************************
This article provides general educational guidance only. It is NOT official exam policy, professional academic advice, or guaranteed results. Always verify information with your school, official exam boards (College Board, Cambridge, IB), or qualified professionals before making decisions. Read Full Policies & Disclaimer , Contact Us To Report An Error
